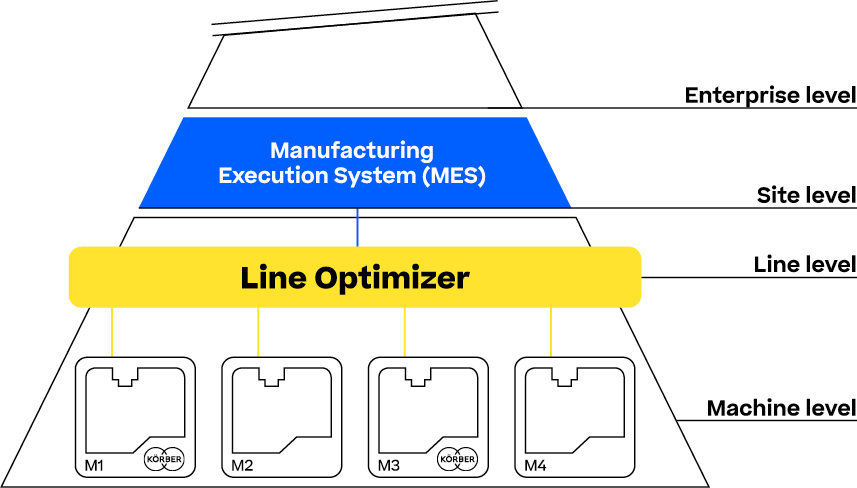
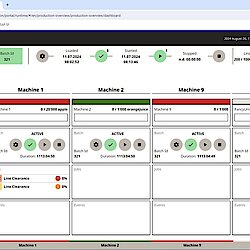
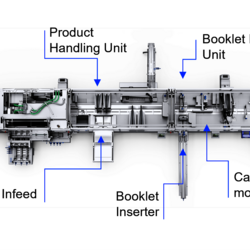
Connect is a powerful module within the Line Optimizer Suite that ensures seamless communication across your production line, regardless of the machine supplier.
By utilizing industry-standard protocols like OPC-UA and ISA TR 88, Connect enables efficient data exchange between machines and MES systems without requiring a Human-Machine Interface (HMI). With PackML as the common language, Connect significantly reduces configuration efforts by up to 70%, simplifies data maintenance and streamlining integration.
Whether it’s managing batch information or synchronizing equipment and workflows, Connect ensures reliable, supplier-independent connectivity, translating and transmitting data effortlessly between machines and higher-level systems.