Solutions
Software solutions
Handling systems
Inspection
Packaging machines
Packaging solutions
Consulting
Services
Ticket System for Machine Services
Software for the pharmaceutical and biotech industries — your path to digital production
Do you want cost-efficient, safe, and flawless production processes? If so, you should control and monitor your production digitally with the help of the worldwide market-leading PAS-X solutions - on premise or cloud-based - for pharmaceutical and biotech companies. Our ecosystem thinking and planning enables us to design your digital production architecture as a modular and open system, keeping you flexible, and evolving accordingly to market developments.
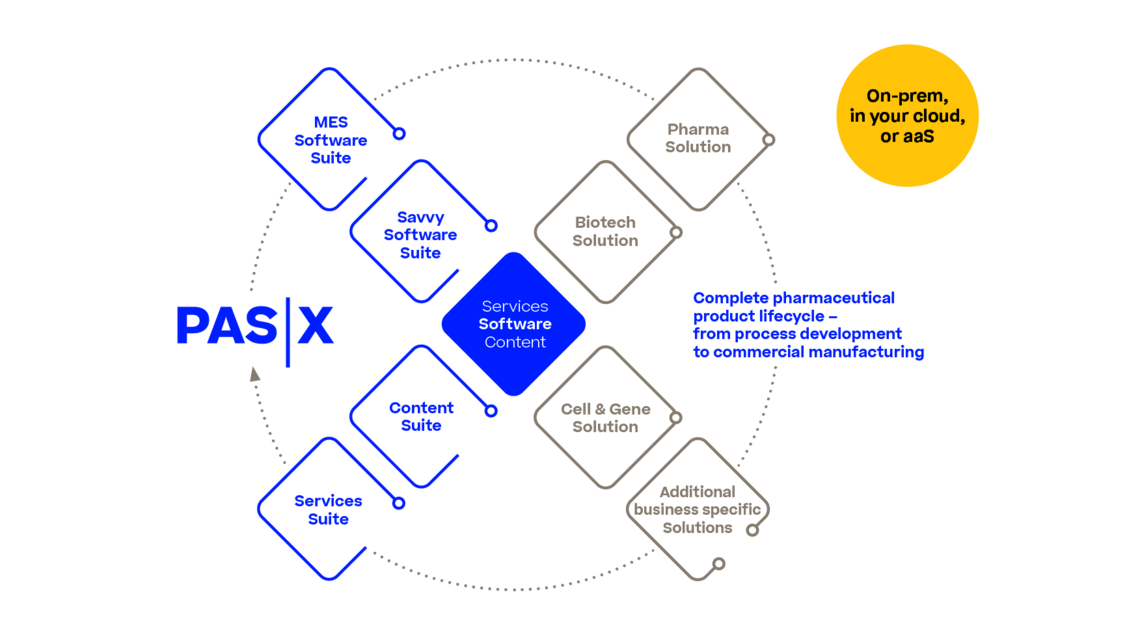
PAS-X Services Overview Cloud-based: Werum PAS-X as a Service Werum PAS-X MES Suite Werum PAS-X Savvy Suite Werum PAS-X Add-ons Werum PAS-X Packaging SolutionWerum PAS-X Content SuitePAS-X CommunityLine management (Lion.Suite) PAS-X Track & TraceWerum PAS-X Cell & Gene SolutionPAS-X BiopharmaSmart packagingPartnersPAS-X K.AI


Werum PAS-X Cell & Gene Solution
As a provider of cell therapies, you face extraordinary challenges due to the highly complex nature of the production processes for personalized medicine. Werum PAS-X MES enables you to control your production process for cell and gene therapies digitally and automatically. This makes your production operations faster and safer.
About us
We are your contact when it has to do with pharmaceutical products. As a leading system supplier, we advise you on everything from development through manufacturing and inspection all the way to the packaging of your products.