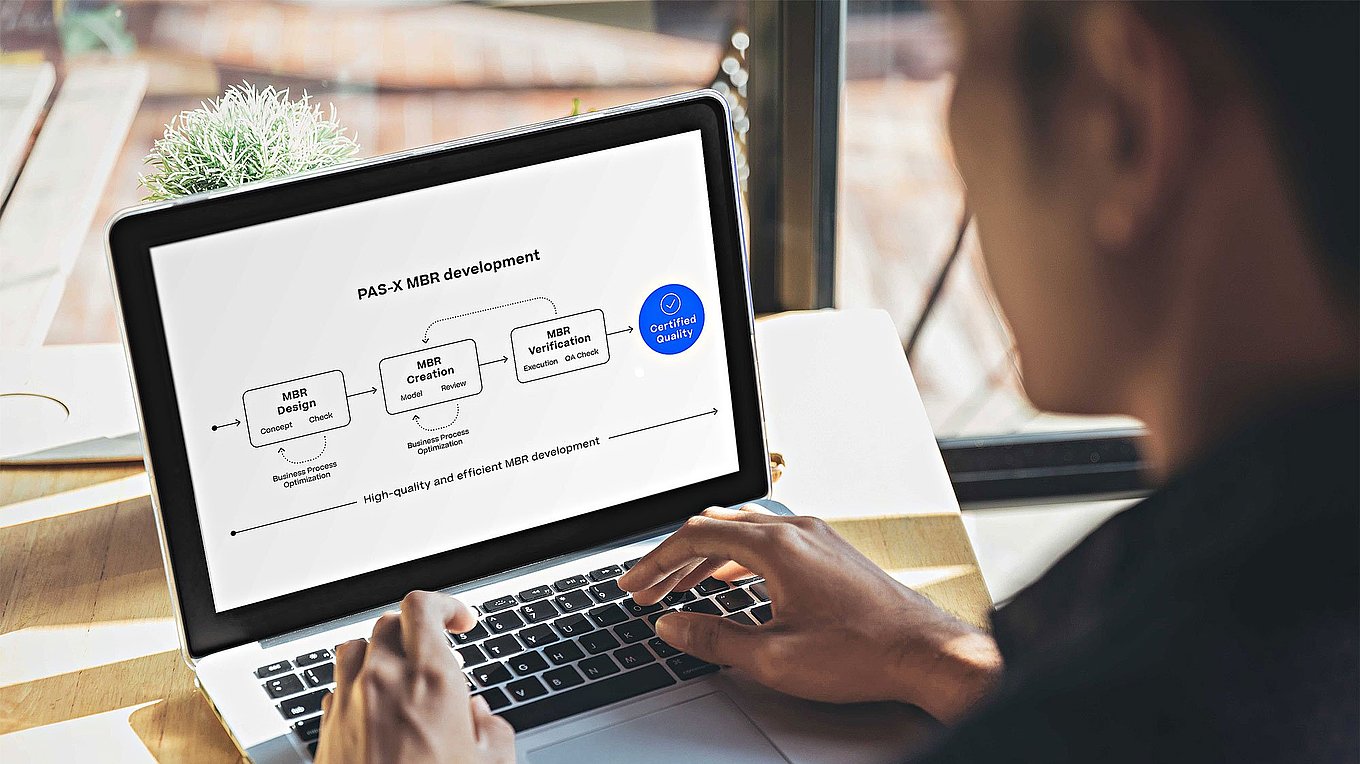
Ensuring the quality of MBR design is critical to successful MES, and, as such, the end-goal of creating high-quality MBRs must guide the entire production and review process.
Successful MBR design requires a structured approach:
Production process analysis
The production processes to be modeled in MBRs should be analyzed. This includes taking into account the requirements of stakeholders (e.g. production and QA), and the boundary conditions of other systems (e.g. ERP order structure). The result of this analysis will include the production process, structured into MBRs (e.g. bulk production, packaging), its unit operations (e.g. weighing, granulation, filling) and process steps. Such steps may include ‘assemble granulator,’ ‘granulation run,’ ‘post-run-activities,’ or ‘yield operations.’