To support the implementation of the already qualified Werum PAS-X MES system, our team of experienced validation consultants can support you with its validation. To ensure that this is done quickly and effectively we offer you our predefined validation documentation package for the validation of your MES solution.
The validation documentation package offers templates with pre-filled information that can be used as the basis for all needed validation documents. Our services range from turn-key to individual support. This offering can further reduce the overhead needed for the validation process of the MES.
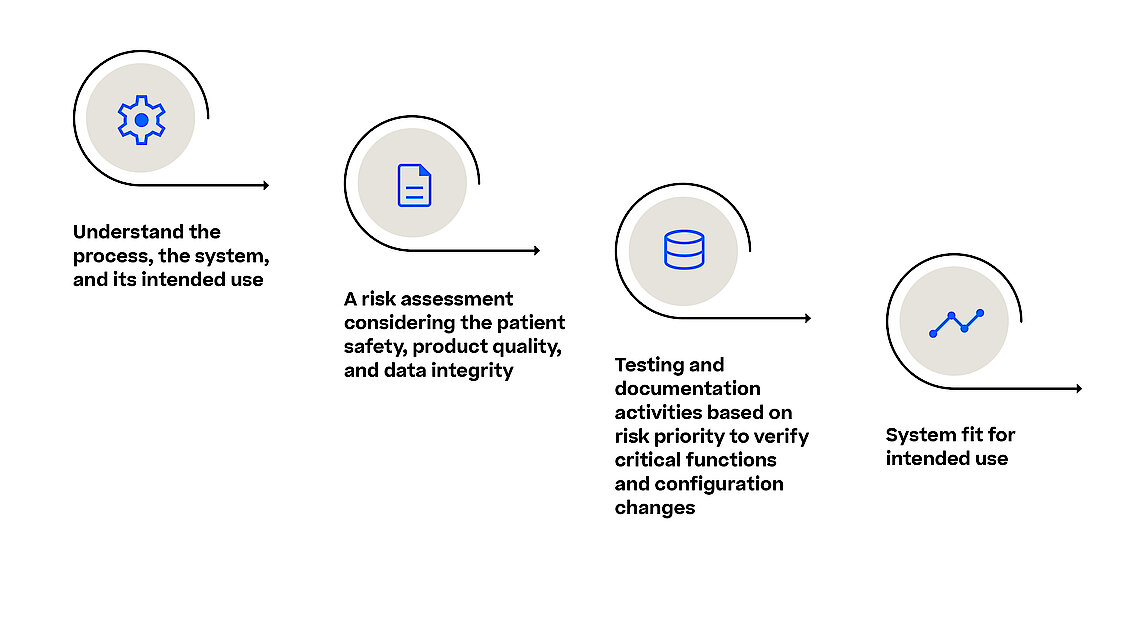
We have aligned our recommended best practice validation strategies, validation documentation package, and project methodologies with the CSA guidance. We increasingly deliver our projects with the help of automated testing for customer specific system changes. In addition, we recommend the use of a risk-based approach and leveraging the supplier qualification testing to reduce the need for functional testing by the regulated user and reduce their own testing and documentation efforts. The validation approach already has been adapted to the requirements of PAS-X MES and the typical processes of the pharma, biotech, and advanced medicine sectors.