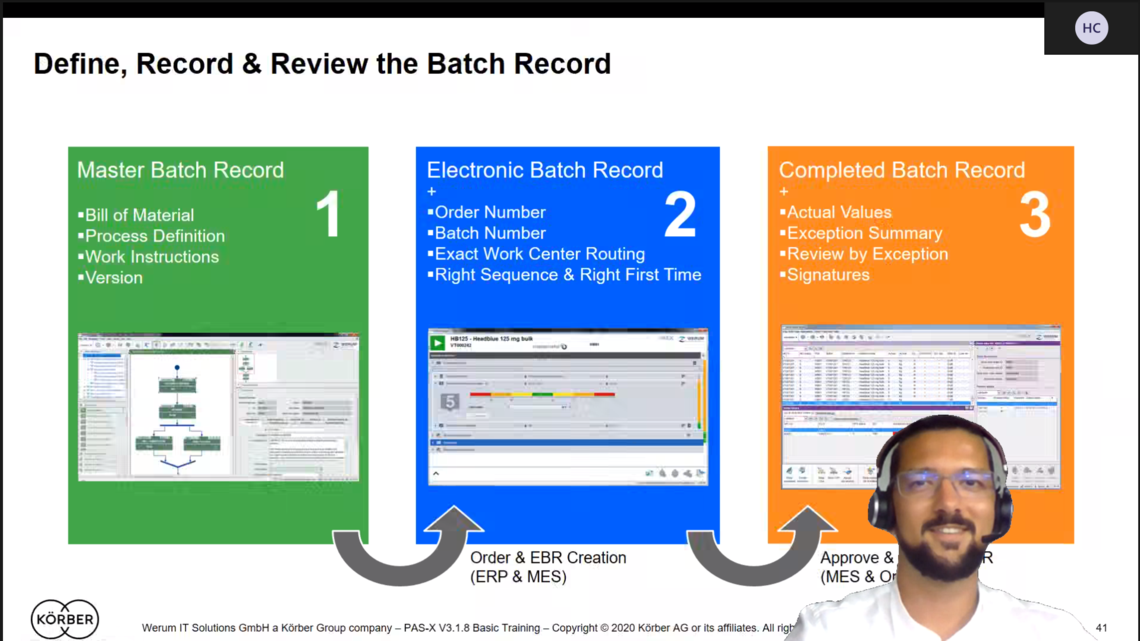
MBR design is the turning key
Another challenging task is the MBR design: creation of the product batch records (recipes) in Werum PAS-X MES is much more than just a transfer of the paper specification 1:1 into the system. This is because it’s not a simple “paper on glass” concept – it’s about optimizing and improving the manufacturing processes for the operators on the shop floor, while having a strong user guidance via clear electronic work instructions and workflow enforcement to achieve “Right First Time”. To design the workflow and instructions including all process parameters and values, a close collaboration with the process owners, subject matter experts and operators is required to get a good understanding of the production process to identify the improvement possibilities, which can be automation of work steps and their documentation.
The big advantage for our customers especially during remote sessions where the consultants cannot see the business process and visit the manufacturing site is that our consultants have an excellent process knowledge and experience of MBR design within the pharma industry. This makes it easier to design an MBR from hundreds or even thousands kilometers away. Additionally to our design guidelines, which guarantee an effective design structure, we use our PAS-X process libraries content packages. These are already predefined recipe building blocks representing the entire process and its sub processes, and designed based on our extensive industry expertise and experience, hence speeding up the recipe creation process for our customers. As a result, this enables seamless manufacturing and fast delivery of life-saving medicines to the market.
Overall, we tackled the challenges coming with the “new normal”, developing, adjusting and improving ourselves, working together with our customers to ensure a smooth implementation and operation of Werum PAS-X MES at their production sites. We are delivering the difference in pharma – no matter if on-site or remote.